Which of the Following Has the Lowest Freezing Point
1 Al2SO43 2 C6H12O6 3 KI 4 K2SO4. A pure H₂O B aqueous glucose 060 m C aqueous FeI₃ 024 m D aqueous KF 050 m E aqueous sucrose 060 m D aqueous KF 050 m Which of the following liquids will have the lowest freezing point.
Solved Which Of The Following Has The Lowest Freezing Point Chegg Com
Answer D h2 The lo.

. Which of the following liquids will have the lowest freezing point. Experts are tested by Chegg as specialists in their subject area. Solved Which of the following has the lowest freezing point.
If seawater is an aqueous solution of sodium chloride calculate the molality of seawater. Compared to the freezing point and boiling point of water at 1 atmosphere a solution of a salt and water at 1 atmosphere has a. A R-11 b R-12 c R-22 d Ammonia.
A 015M glucose b 030 M sucrose c 015 M NaCl d 030 M CH3COOH. Which of the following has the lowest freezing point. Which of the following solutions have lowest freezing point.
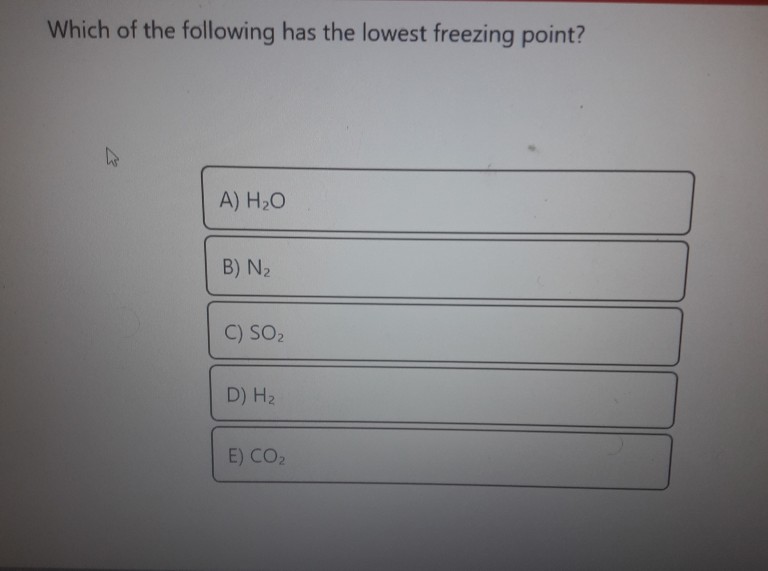
Assume the value of iis 2 for NaCl and. Which of the following solutions have lowest freezing point. AHO B N 2 C SO DH E COZ.
A pure H2O B aqueous CoI2 0030 m C aqueous NaI 0030 m D. Freon 22 has the lowest freezing point temperature from all of. Which of the following refrigerants has lowest freezing point.
View the full answer. Which one of the following 006 M aqueous solutions has lowest freezing point. Which will give the lowest freezing point of water.
Mixing 176 kg of ethylene glycol commercial antifreeze C2H6O2 with 600 gal of water lowers the freezing point to 100 F. The material with the lowest freezing point is helium. Helium has the lowest freezing point.
01mCaI2 will have the lowest freezing point followed by 01mNaCl and the highest of the three solutions will be 01mC6H12O6 but all three of them will have a lower freezing point than pure water. Ective molality is the highest in the group and therefore will have the lowest fp the highest bp and the maximum osmotic pressure. Add your answer and earn points.
Which of the following 01 M aqueous solutions will have the lowest freezing pointA. Aqueous sucrose 075 m d. 1m NaCl 15m CH3CH2OH 07m AlCl3 12m CsNO3.
A 10 molkg NaClaq b 10 molkg CaClaq c 05 molkg NaClaq d 05 molkg CaClaq 115. Which of the following solutions has the lowest freezing point and the highest boiling point. Which of the following would have the lowest freezing point.
I know the answer is d because our professor told us so but he did not explain why or how I would calculate to find the answer could you please explain. The zero point energy of a helium system is too great to allow freezing. Amelia4152 amelia4152 Remember the greater the concentration of particles the lower the freezing point will be.
Under typical pressures it does not freeze at all even at temperatures approaching absolute zero. AHO B N 2 C SO DH E COZ. A 01NaCL b 001 M NaCLc 1M NaCLd 0001M NaCL.
Aqueous lif 065 m b. 0102 m sucrose 0050 m NaCI b. What is the new boiling point assume the original boiling point is 100C if you dissolve 55 g NaCl molar mass of NaCl is 5844 gmol in 075 kg of HO.
Chemistry questions and answers. 022 100 points The freezing point of seawater is about - 185 C. The k f for water is 186 K m.
We review their content and use your feedback to keep the quality high. Which of the following solutions will have the lowest freezing point. The reasons why are dictated by quantum mechanics.
Which of the following has the lowest freezing point. Higher freezing point and a lower boiling point. Freezing point depression is a colligitive property of water that depends in part on the number of ions.
Which of the following refrigerant has the lowest freezing point. Lower freezing point and a higher boiling point. Simply find the solution with the highest value of mxi a.
Which of the following liquids will have the lowest freezing point. Lower freezing point and a lower boiling point. The solution of BaOH2 will have the lowest freezing point.
This is because it dissociates into 3 ions one Ba and 2 OH which is more than any of the others. Among the given options R-22 has lowest freezing point. Which of the following has the lowest freezing point.

Which One Of The Following Solutions Has The Lowest Freezing Point

Solved Which Of The Following Has The Lowest Freezing Point Chegg Com
Comments
Post a Comment